GREENER ENERGY
Harnessing light and air for cleaner processes
GREENER ENERGY
Harnessing light and air for cleaner processes
From photosynthesis in plants, to every breath we take and every thought we think, life is animated by complex chemical reactions that pass electrons back and forth between molecules.
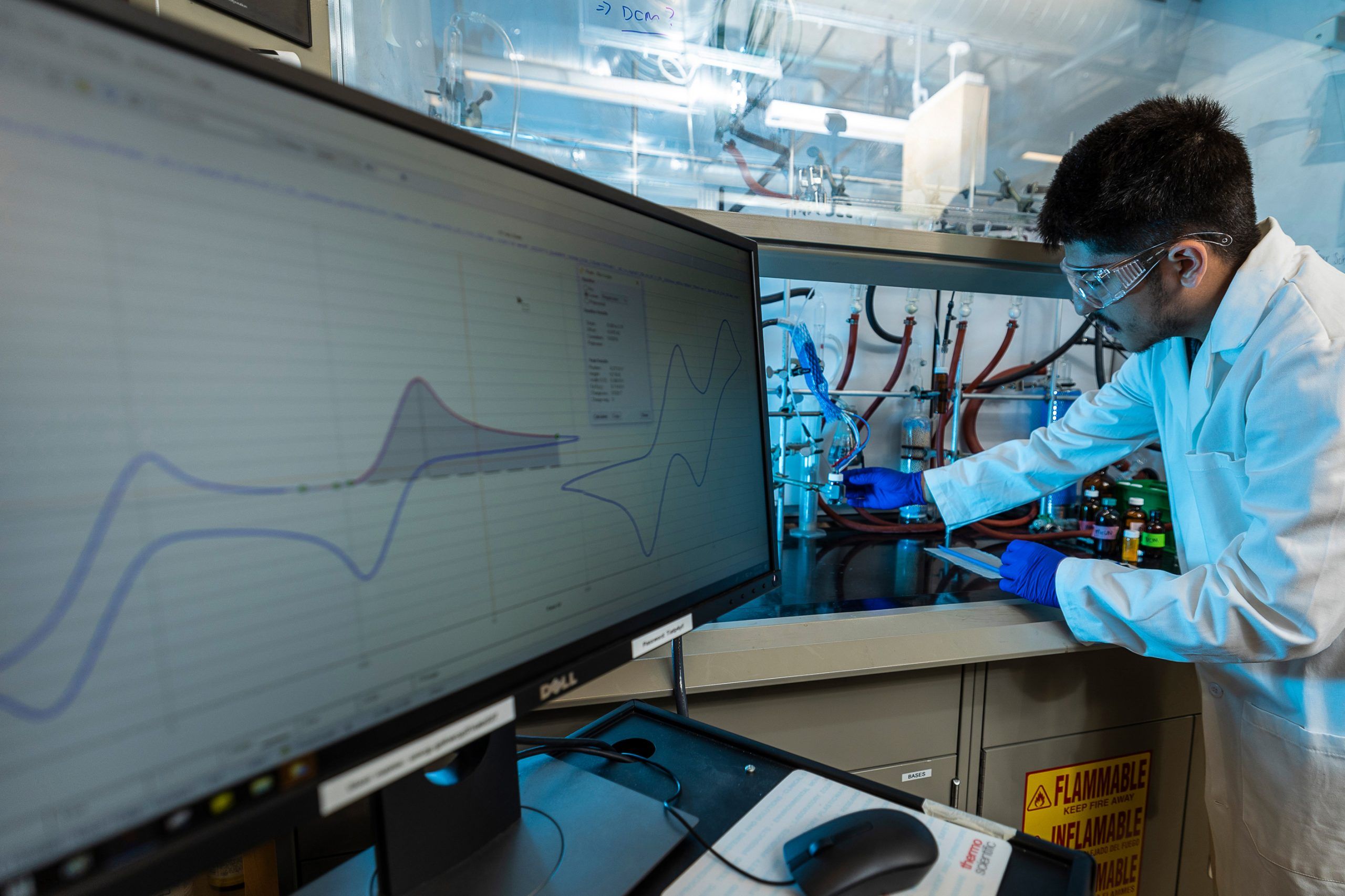
Dr. Shabnam Hematian, Bernard-Glickman Dean’s Professor in the UNCG Department of Chemistry and Biochemistry, has focused her research on the metals — iron, copper, and others — that are foundational to these processes.
“The cool chemistry that happens in your body – metabolism of drugs or hormone biosynthesis – all of them are governed by a series of iron proteins,” she says.
Inspired by nature, Hematian is working to understand how light and air, along with common metals, could catalyze industrial reactions, to design more efficient and environmentally friendly technologies.
Early-career scientist Shabnam Hematian is bringing a natural process lens to energy research. With a $250K NSF grant, she’s exploring how light and air might help catalyze industrial reactions for more environmentally friendly technologies – and pursuing her passion for mentoring.
Understanding how they could be harnessed to drive electron transfers in industry could change the design of high-capacity fuel cells, for example, and allow companies to create them without more expensive and more toxic metals such as platinum or ruthenium.
“I’m looking at where and how electron flows happen. It can be in a battery or it can be in your mitochondria.”
Photos 1, 3, 4: Hematian and postdoc Firoz Khan load a sample into the x-ray crystallographer at the JSNN. The machine allows them to determine the 3D molecular structures of their photocatalysts. | Photo 2: Local high school students Fully Porter and Nicholas Alejandro use a nuclear magnetic resonance spectrometer in Sullivan Science Building to determine the molecular composition of a sample. The work is part of their Draelos Scholars research experience in Hematian’s lab.
Inspired by nature
As a researcher with expertise in both synthetic chemistry and light-driven biological reactions, Hematian brings a natural process lens to industrial questions.
“Nature started working and figuring out how to do transformations with just the bioavailable elements,” she says. “We don’t have any enzymes that have ruthenium in them, but we have a lot of enzymes that have iron in them.” Those enzymes may provide models for more environmentally friendly reactions.
Light and air are abundant and, Hematian explains, light’s color, intensity, and other properties can be precisely controlled. Thus, it has the potential to become a sensitive tool to drive chemical reactions. In the future, she says, adjusting the color of light might allow scientists to activate one part of a molecule but not another.
In the spring of 2022, Hematian was awarded a prestigious two-year, $250,000 grant from the National Science Foundation’s LEAPS initiative, which is designed to support promising mathematicians and physical scientists early in their careers.
With the funding, Hematian is developing new ways to transform carbon-based materials into oxygenated materials. These kinds of reactive materials, like ethanol, are valuable in industry, but making them currently requires harsh chemicals, high temperatures, and high pressure.
“They’re very expensive types of reactions,” says Hematian. “We want to develop catalysts that can absorb light to give us the energy for these transformations instead.”
In another example of applying natural inspiration to 21st century challenges, Hematian is looking at molecules found in certain fungi that could be useful in liquid batteries.
The work is emerging from a collaboration with Patricia A. Sullivan Distinguished Professor of Chemistry Nick Oberlies. The natural products chemist and his team assess compounds found in fungi for medicinal applications, but Hematian has recognized the potential in some of their findings for electrochemistry.
Liquid – or “flow” – batteries store more energy than current solid state battery technology and are the wave of the future, she says. “Many startups are currently working on figuring out which compounds are most efficient and durable for use in them.”
Photos: Undergrad Marcos Tapia conducts research in Hematian’s lab. In his sophomore year, the first-gen student has already co-authored a scientific article and mastered an in-demand electrochemistry technique.
Catalyzing learning
The NSF LEAPS grant also supports another of Hematian’s interests: mentoring students from a variety of backgrounds.
She’s recruiting undergraduates, community college students, and even some high school students to work in her lab alongside graduate students. She also wants to involve home-schooled students.
“I’m hoping this is going to connect what they learn in their textbooks to life – now they’re doing it in the lab.”
The goal is to give students first-hand experience that not only reinforces classroom learning, but also helps them understand how original research is actually done. “We may get results we don’t understand at the beginning. How can we go about handling that?”
The work will expose the students to researchers and science-based career opportunities.
As a researcher who has benefited from strong mentorship and similar opportunities, Hematian says it’s important to her to continue that tradition.
“I’m very invested in passing the knowledge to the next generation,” she says.
“That’s my passion: to influence people and the future.”